Boron Empty P Orbital
Boron's empty p orbital in bf3 Ethene molecular orbital diagram Boron for undergraduates
Back bonding is possible in BF3 even though it has no vacant d orbital
Boron electron configuration Electron orbitals (a-level) Overlap orbital boron chemistry structures resonance represented also
Which of the following species is not electrophilic in nature:a
Organic chemistry 03: bonding14.4: lewis acid-base complexes and molecular orbitals Boron figure orbital interactionsBoron configuration electron orbital diagram 3a find carbon because.
Boron empty electrophilic presence hydrogen orbitalOrbitals electron orbital atomic pz py shell energies Bond orbitals molecular bonding pi ethylene chemistry organic mo atomic diagrammed follows understandOrbital molecular diagram ethene orbitals bonding organic chemistry khan atomic mo borane academy bonds sigma dienophile substituents diene depend.

Bf3 bonding orbital vacant possible boron
Bf3 orbital orbitals boron bond hybridization bonding electron valence trifluoride libretexts hyb chemBack bonding is possible in bf3 even though it has no vacant d orbital Boron configuration electronEmpty orbital electrons orbitals electron pairs bond oxygen interacting interaction atom carbocation however resembles.
Lewis molecular orbitals complexes libretextsChemistry: factors affecting orbital overlap, boron halides Empty p-orbitals interacting with electron pairs — organic chemistry tutorHow to find the electron configuration for boron.


14.4: Lewis Acid-Base Complexes and Molecular Orbitals - Chemistry

Chemistry: Factors affecting orbital overlap, boron halides

Organic chemistry 03: Bonding - atomic orbitals and molecular orbitals

Boron's empty p orbital in BF3 | Student Doctor Network

Which of the following species is not electrophilic in nature:a
BORON FOR UNDERGRADUATES
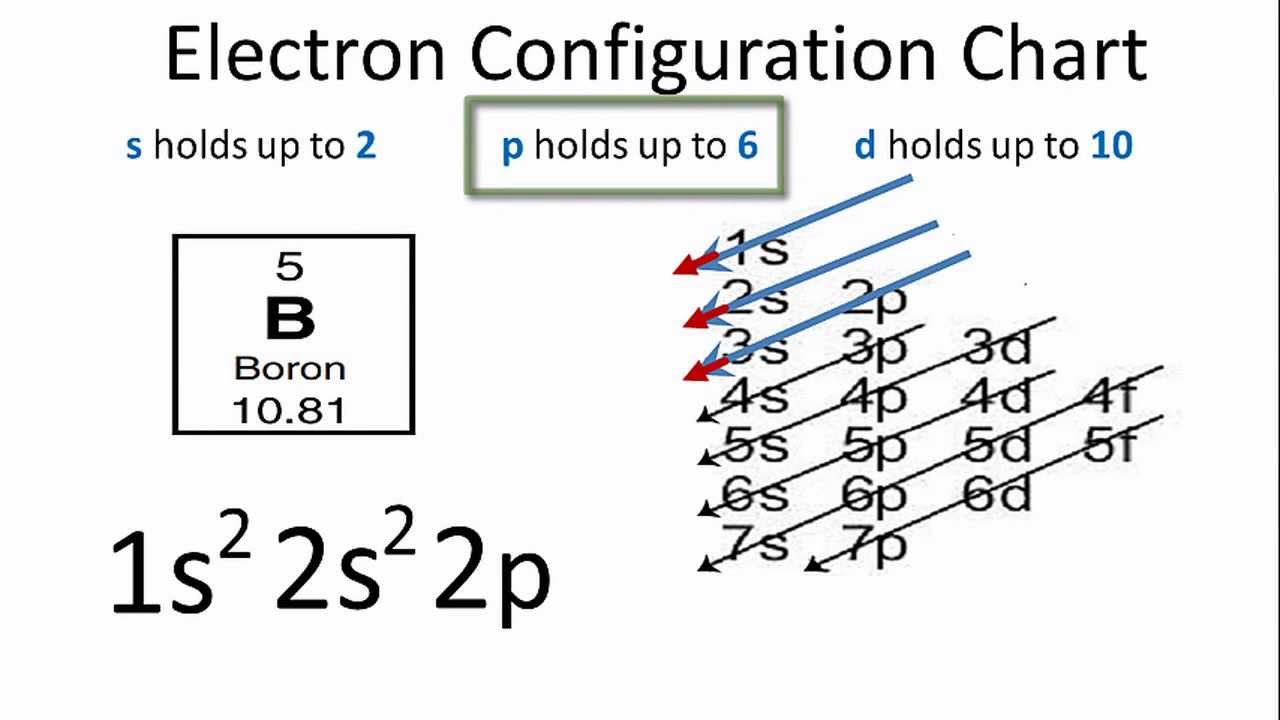
Boron Electron Configuration - YouTube

Empty p-Orbitals Interacting with Electron Pairs — Organic Chemistry Tutor

Electron Orbitals (A-Level) | ChemistryStudent

Back bonding is possible in BF3 even though it has no vacant d orbital